|
A regular period is a vital sign representing our reproductive and general health (and let's you know about egg health!). About 14 days before your period arrives you may just be ovulating but don’t take it for granted that you are ovulating just because your period arrived. In fact, research suggests that ovulation does not take place in 16-37% of menstrual cycles. [1] Just as our period arriving regularly is a sign of our health, so is the presence of ovulation. Let’s take a look at how we ovulate, what can impact ovulation, and signs our ovaries are doing their job. Back to Basics: Menstrual Cycle 101Our ovaries contain eggs that develop inside follicles. Follicles are like little water baths for the eggs and the contents of this fluid that our eggs sit in impacts the quality and health of our eggs. If this fluid contains a good balance of antioxidants and anti-inflammatory compounds, and the right hormone and nutrient levels it will nurture our eggs and protect our follicles from damage. Most research supports that we are born with all the eggs we will ever have. These eggs sit hibernating, producing just enough energy to survive. When they are finally called on to grow and be released, the maturation process takes a few months. Nurturing your eggs before and during this maturation phase provides the best opportunity for producing healthy eggs. While 15-20 follicles will be maturing at the same time, only one follicle is chosen to reach the final stage and become the dominant follicle. If the right conditions are provided for this follicle, this will be the one to release an egg that menstrual cycle. The egg will then make its way into the fallopian tube and if it meets sperm, this is when fertilisation may take place. Hormone Harmony The hormone Follicle Stimulating Hormone (FSH) drives the development of follicles, just as the name says. As the follicle develops, oestrogen is released from cells that surround the egg (granulosa cells that are part of the follicle). When oestrogen levels rise enough, this triggers a surge in Luteinising Hormone (LH). LH comes from the pituitary, a small gland at the base of our brain. This hormone triggers enzymes that breakdown the follicle and ovarian wall to let the egg out. It is an incredible process and has been captured on camera once during a surgical procedure. If you want to see the photos of a follicle releasing an egg from the ovary you can see these at the New Scientist here. Once the follicle has ruptured to release the egg, the remnants of the follicle then get to work and produce progesterone. Progesterone is essential for sustaining a pregnancy if the egg that has just been released gets fertilised and implants into the uterus. When Ovulation Gets DisruptedThe process of growing a follicle and successfully releasing an egg relies on good communication pathways from the brain to ovary. A region of our brain called the Hypothalamus sends out signals to our pituitary at the base of our brain, which then sends out signals to our ovaries. Parts of the body that communicate in sequence can be called an axis and ovulation relies on a well-functioning Hypothalamic-Pituitary-Ovarian (HPO) axis. When ovulation does not place as it usually should, 85% of these failures may be attributed to disruptions with the HPO axis. What Disrupts the HPO Axis? Elevated Testosterone: High levels of androgens, such as testosterone, can impact the balance of hormones and the ratio of LH to FSH. The ratio of LH to FSH is usually 1 to 2 in women without identified fertility issues, but in conditions such as PCOS this ratio can be as high as 3 leading to disruption of ovulation. Insulin Resistance: sex hormones may be carried around the body by specialised proteins. Insulin resistance is related to a decrease in the production of these sex-hormone-binding proteins which may contribute further to raised levels of androgens. Controlling blood sugar and managing insulin resistance is essential for healthy ovulation. High Inflammation: Higher levels of inflammation levels during the phase of follicle development have been associated with a lack of ovulation (or anovulation) [2]. Inflammation is necessary for ovulation but if levels are too high, this disrupts follicle development. This inflammation underlies unruptured follicle syndrome which is a common cause of infertility with endometriosis [3]. Elevated Lipopolysacharide (LPS): LPS can be found in the cell wall of particular bacteria. With an increase in intestinal permeability, systemic levels of LPS can rise and this can further drive inflammation. High levels of LPS (which are seeing in PCOS, endometriosis, unexplained infertility and male factor infertility) disrupt the surge in LH and have been associated with premature ovarian insufficiency, reduce egg quality and early pregnancy loss. This is why building our microbiota diversity for a healthy gut optimises our reproductive health. Low Energy Availability: If we are not providing our body with enough energy to meet the needs of our daily activities and basal metabolic rate, this may lead to menstrual cycle disturbance. The amount of energy we may be missing out on in our diet is predictive of menstrual disturbances. A greater energy deficit contributes to greater disruption of ovulation. The Gut-Ovulation ConnectionWhy would my gut play a role in ovulation? All the elements that can disturb our HPO listed above are all linked to the health of our gut microbiota. Depending on the health of our gut microbes they can either promote or dampen inflammation. They are also the source of the endotoxin LPS. If our gut microbes are out of balance our intestinal barrier becomes more permeable, leading to an increase in circulating LPS levels. When our gut breaks down proven prebiotic compounds they produce short chain fatty acids like Butyrate. These compounds have been shown to improve glucose homeostasis. Higher circulating short chain fatty acids also leads to better insulin sensitivity (which our eggs love) [4]. In women with PCOS, nurturing the gut microbes reduces androgen levels, inflammation and LPS to restore ovulation. If you have unexplained anovulation and do not have PCOS, it is likely that your gut microbes are different from women that are ovulating regularly. Research shows that anovulation is associated with a greater abundance of Prevotella-enriched microbes that promote inflammation and a reduction in butyrate producing bacteria [5]. Endometriosis is also associated with more inflammatory promoting bacteria and this can be a cause of menstrual pain. Nurture the diversity of your gut microbes will have huge benefits for healthy ovulation, and healthy eggs! Signs of OvulationThe gold standard way to tell if you are ovulating is to have repeat transvaginal ultrasounds to track follicle growth and rupture. This is definitely not a very accessible way of tracking ovulation. So how else can you get some clues to know if ovulation is taking place? Temperature Tracking This method of ovulation tracking requires you to monitor your temperature daily each morning so you can detect changes across your menstrual cycle. A small rise in your morning body temperature of 0.3 degree C / 0.5 F my you let you know that ovulation has occurred. Once our follicle ruptures to release an egg the left-over follicle produces progesterone which tells our higher brain centres to set a higher body temperature. While an increase in temperature may indicate ovulation, many other things also affect our temperature such as alcohol, sleep disruption and stress. Also, for 20% of women they may still ovulate but not have a change in body temperature [6]. Pee on a Stick Measuring urine LH to determine when levels ‘surge’ is another method that may indicate ovulation. The rise in LH typically occurs 36 hours before ovulation. The type of rise in LH is not necessarily a rapid surge for all women though so tracking this can be difficult. It is also possible to have an LH surge but no ovulation, as seen in unruptured follicle syndrome mentioned above. Cervical Mucus Monitoring Under the influence of oesterogen, the water content of our cervical secretions increases and mucin content decreases. If your cervical mucus is very wet, stretchy and resembles raw egg white then ovulation is very close! This method of ovulation tracking is 74% accurate when compared to tracking ovulation via the gold standard of transvaginal ultrasound. Position of your Cervix As you get closer to ovulation, your cervix rises and becomes softer. Keep Track of Your Vital SignsSo are your ovulating? Keep track of the health of your menstrual cycle (ovulation, cycle length, symptoms, bleeding etc) and continue to nurture your gut microbiota for optimal egg healthy and uterine receptivity. If you would like support to get your cycle back on track, work with us to implement evidence based strategies for pregnancy success! References
1. Prior, J.C., et al., Ovulation Prevalence in Women with Spontaneous Normal-Length Menstrual Cycles - A Population-Based Cohort from HUNT3, Norway. PLoS One, 2015. 10(8): p. e0134473. 2. Lorenz, T.K., C.M. Worthman, and V.J. Vitzthum, Links among inflammation, sexual activity and ovulation: Evolutionary trade-offs and clinical implications. Evol Med Public Health, 2015. 2015(1): p. 304-24. 3. Mio, Y., et al., Luteinized unruptured follicle in the early stages of endometriosis as a cause of unexplained infertility. Am J Obstet Gynecol, 1992. 167(1): p. 271-3. 4. Muller, M., et al., Circulating but not faecal short-chain fatty acids are related to insulin sensitivity, lipolysis and GLP-1 concentrations in humans. Sci Rep, 2019. 9(1): p. 12515. 5. Sasaki, H., et al., Distinctive subpopulations of the intestinal microbiota are present in women with unexplained chronic anovulation. Reprod Biomed Online, 2019. 38(4): p. 570-578. 6. Barron, M.L. and R.J. Fehring, Basal body temperature assessment: is it useful to couples seeking pregnancy? MCN Am J Matern Child Nurs, 2005. 30(5): p. 290-6; quiz 297-8. In the journey towards parenthood, in-vitro fertilization (IVF) has become a lifeline for many struggling with infertility. But preparing for IVF success goes beyond just medication timings and hoping for the best. It requires a comprehensive understanding of the process and a strategic approach to maximise your chances of achieving a successful outcome. In this blog, we will delve into some elements that can positively impact your IVF journey, providing you with valuable insights and practical tips to help you navigate this complex journey. Whether you are considering IVF for the first time or have undergone multiple rounds, this blog will equip you with knowledge and tools you need to boost your chances of success. So, let's dive in and discover how you can take proactive steps to prepare for IVF success and increase your chances of starting or expanding your family. Understanding the IVF Process and WHY you are Undergoing Fertility TreatmentIf you have had trouble conceiving the next steps may involve assisted reproductive treatments. These may include ovulation induction, intrauterine insemination (IUI), in-vitro fertilisation (IVF) or intracytoplasmic sperm injection (ICSI). The best predictor of fertility success relates to the quality of egg and sperm used to make an embryo. This applies if you are conceiving without intervention, or with assisted reproductive treatments. Identifying why you may be having difficulty conceiving is an important first step in supporting your preparation for treatment. Is your period irregular or absent? You want to get to the bottom of why this is occurring and address the cause. Regular menstrual cycles are a great indicator of health and egg quality. Are there Male Factors? If there are parameters in the semen analysis that are low or not in normal range IUI / IVF / ICSI are not going to improve sperm quality- but you can! Male factors are critical for conception and even with a normal semen analysis there is still work to be done in preparing. Sperm DNA damage (not detected in a semen analysis) adversely affects embryo quality and implantation. An egg can repair sperm but it takes a lot of energy and this requires our energy producing powerhouses in our eggs, our mitochondria, to work overtime. If the mitochondria content in our eggs is reduced, or the mitochondria just aren’t producing enough energy, our developing embryos run out of steam. If you have PCOS, endometriosis, are not ovulating regularly or an imbalance in your gut microbiome you are likely to have less mitochondria, and these may not be producing as much energy. Even without any male factors identified, sperm needs to be looking great to help out our eggs! Do you have an Autoimmune condition such as endometriosis, PCOS, adenomyosis, Thyroid condition, Rheumatoid Arthritis, IBD, SLE, Coeliac Disease? These conditions impact our gut microbiome. Supporting your immune system to reduce inflammation is key to change the follicular fluid environment and support your developing eggs, and implantation. Have you been told you have ‘Unexplained Infertility’? Do you know if you have a healthy and diverse gut microbiota? Do you have elevated levels of systemic inflammation and lipopolysaccharide (LPS)? Do you have a high percentage of DNA fragmentation? Are you micronutrient deficient? Is your exercise supporting your fertility? Do you need to increase your microbiome diversity? Are you getting enough fibre in your diet? Is your diet adequate to support your activity? Are you being exposed to environmental toxins regularly? Are you experiencing or recovering from burnout? Is your sleep supporting recovery and restoration? Is your sympathetic nervous system overwhelmed? The list goes on as there are many factors that impact fertility that are not captured in routine testing. With our research science background and evidenced based practice you are likely to uncover strategies to support why it has been more difficult to conceive or have a successful pregnancy. Time Frame to PrepareIVF does not change your egg or sperm quality, or uterine receptivity. Improving egg and sperm quality BEFORE fertility treatment will provide you with the best chance for success. Our eggs mature over 120-290 days and sperm may take around 74 days to be made. The environment our eggs and sperm mature in over these time frames will impact their quality. Planning to focus on your health for a minimum of 3-12 months prior to conception may save you expensive cycles of treatment and reduce the emotional toll. A bonus is that our lifestyle is capable of altering the next generation through epigenetics so we also get to give our future bub the best start in life! Remember that it takes two - healthy eggs AND sperm are the building blocks for success. The Foundation: Your Gut Microbiome Did you know that the billions of bacteria residing in your gut and the compounds they produce- collectively known as the gut microbiome - play a crucial role in regulating various aspects of your reproductive health? From hormone production to nutrient absorption, these helpful bacteria heavily influence your fertility. A healthy gut microbiome can positively influence reproductive function and IVF outcomes. To optimise your chances of IVF success, it is important to focus on supporting a diverse and balanced gut microbiome, and this will support a healthy uterine microbiome. A healthy gut microbiome is essential for maintaining hormonal balance, which is key for successful conception. The gut microbiota interacts with your body's endocrine system, helping to regulate the production and metabolism of hormones such as oestrogen and progesterone. Imbalances in these hormones can disrupt the menstrual cycle and make it difficult to conceive. Our gut microbiome also plays a role in nutrient absorption. Without proper nutrient absorption, your body may lack the essential vitamins and minerals needed for reproductive health. This can impact the quality of eggs and sperm, making it more challenging to conceive. To nurture your gut health and enhance fertility, it's important to focus on nourishing your gut microbiome. This can be achieved through a gut-friendly diet that includes specific nutrients and compounds to nurture beneficial bacteria. There is a strong connection between gut health and fertility, and we wrote the book that Fertility Experts and Dietitians are reading! If you LOVE science, you can read Create a Fertile Gut: A Science Backed Approach to Nurture Your Microbiome for Optimal Fertility, IVF and Pregnancy Success. Exercise and IVFRegular exercise is not only beneficial for overall health but can also improve your chances of IVF success. Exercise can help regulate hormones, reduce stress, improve blood flow to the reproductive organs, reduce inflammation, lower DNA damage and improve mitochondria function. Ultimately these lead to better quality eggs and sperm, and uterine receptivity. It is important to strike the right balance of activity though, and to ensure you are getting the desired adaptations to align with your needs. With our specialist knowledge in exercise and IVF we can determine the optimal balance to support egg and sperm quality for conception. Emotional Preparation for IVF Building your family can be an emotional rollercoaster, especially when navigating infertility and IVF. But remember, your emotional well-being is just as important as your physical health throughout this journey. There's incredible support available! Working with a psychologist specialising in fertility can be a powerful resource to help you manage the emotional ups and downs of IVF. In Australia, a Mental Health Care Plan might even help with accessing a psychologist through Medicare rebates. Am I Ready for IVF?Preparing for IVF success requires a multifaceted approach that encompasses physical, emotional, and lifestyle factors. IVF is a complex journey but the right support and guidance along the way can help you navigate the process, and prepare for the best opportunity for success.
We implement a scientific, evidenced based apporach at The IVF Project. Evaluation is an important part of any planning – even fertility planning. You want to know that the steps you are taking are moving you in the right direction. While you can hold out till another round of fertility treatment or until you are pregnant to know things have worked, you can check in along your journey to conception for signs you are creating a fertile gut. Evaluation does not always have to be about getting more invasive tests to see if your fertility is improving. There can be some simple indicators that your gut microbiota are supporting your fertility and you are getting things back in balance. Here are some questions you may ask yourself and components you could monitor month to month:
If you do wish to have a blood test, semen analysis or Microbiome testing you may like to evaluate how you are progressing by checking:
No matter if you are trying to conceive naturally or are undergoing fertility treatments, you have the power to nurture the quality of your eggs, your sperm, and to create a receptive uterus. Whether you have started treatement or are 5 of more stim rounds in we are here to support you! Our autonomic nervous system controls involuntary processes in our body – in other words – process that are automatic. Our autonomic balance matters as it impacts our fertility. Things like breathing, our heart rate and digestion are regulated by our autonomic nervous system. Even our microbiome is impacted by our autonomic nervous system! We can divide our autonomic nervous system into our “fight or flight” / sympathetic division and our “rest and digest” parasympathetic division. Our parasympathetic nervous system will tend to decrease our heart rate while our sympathetic nervous that dominates during exercise and stress tend to increase our heart rate. The balance between our sympathetic and parasympathetic branches of the autonomic nervous system is impacted by psychosocial environments, physical activity training load, workload, sleep adequacy and diet quality. If we tune into our autonomic balance we can gain insight into how we are coping, adapting and recovering. It might also interest you to know that this balance of autonomic activity is even associated with IVF outcomes and pregnancy success. Let’s dive in to learn more! What is Heart Rate Variability?One tool we use in our research to monitor autonomic balance is heart rate variability. While you may have heard of heart rate, heart rate variability is a bit different. Heart rate variability (HRV) is the variance in time between your heart beats. While your heart rate at rest may be 60 beats a minute, this does not mean that your heart will beat every second, on the second at the same rate. There is some variance in the amount of time between beats and this is what we term heart rate variability. Your heart may have 0.9 seconds between beats and then 1.3 seconds to the next beat. It is variable. When the time between your heart beats becomes less variable, this can be a sign of:
Generally speaking, the more variable the time between heart beats (heart rate variability), the better your health and wellbeing. We are experienced at HRV monitoring and interpretation and have published a few papers in this space. It’s another reason why we love brining the science to optimise your fertility! How do you Measure Heart Rate Variability (HRV)?We use different tools in the research lab to monitor heart rate variability but there is a tool you may have access to at home – a smart watch! While smart watches may not provide the depth of analytics we can investigate, they are a practical alternative to check in on your HRV and monitor trends over days, weeks and months. On an apple watch you will be able to view heart rate variability and this can also be available on certain Samsung smart watch models. On a Garmin the Body Battery has an input of HRV so this can also provide insight. Some Fitbit devices will also provide a heart rate variability metric. It is important to consider when you are wearing the device as capturing data only during exercise sessions will need to be interpreted differently from if you are capturing data as you sleep. This is where we can help! How do I Improve my Heart Rate Variability (HRV)?Heart rate variability is impacted by all the things that impact our autonomic nervous system. This includes psychosocial environments, physical activity training load, workload, sleep adequacy and diet quality. By optimising nutrition, exercise, sleep and mental health and wellbeing, heart rate variability improves. Taking an individualised approach, heart rate variability insights enable your training to be tailored for better adaptations and fertility outcomes. We can monitor how your body responds to different exercise sessions and intensities and adjust recovery periods between sessions appropriately. Heart rate variability also provides insights into diet quality and how you are coping with the emotional load of fertility treatments. By monitoring your heart rate variability may even get insights into the onset of a cold or illness before other symptoms appear! The Science of HRV for IVFThere are many tools that provide us with insight into how your fertility is tracking, and one of these we use is heart rate variability. If you are interested in diving into how your autonomic nervous system is functioning and how to optimise it with science backed strategies, come and work with us!
Referenes
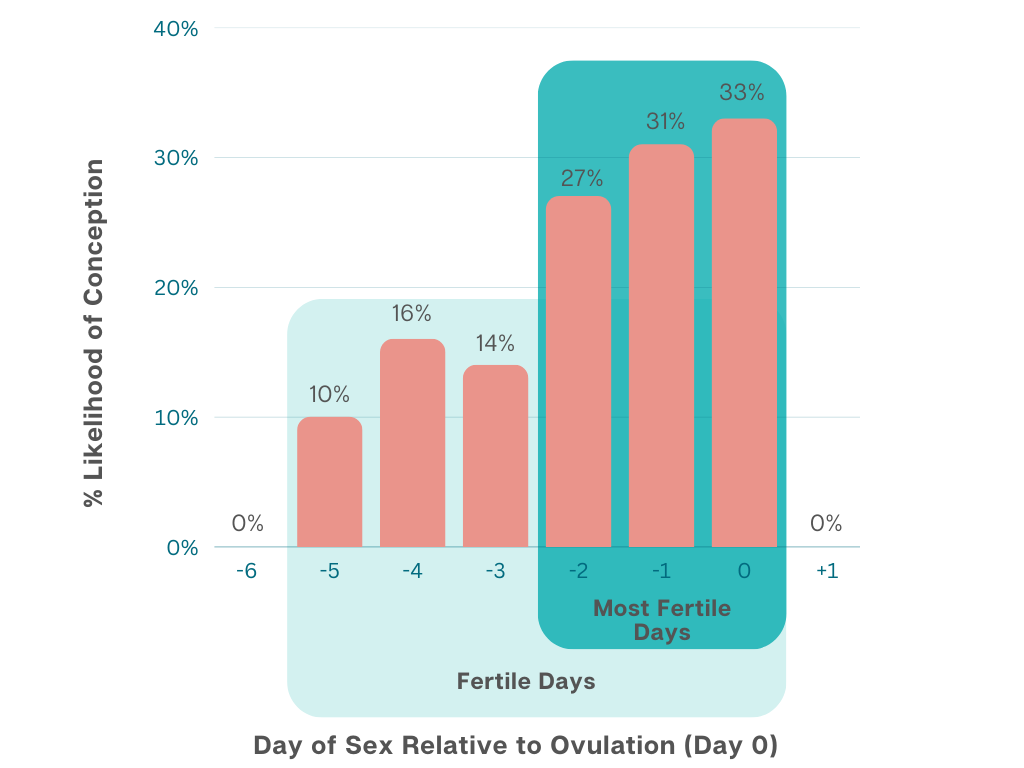
An et al, J Assist Reprod Genet. 2013 Wu et al, PLoS One. 2018 Young et al, Behav Pharmacol. 2018 You might be thinking about growing your family, already trying to conceive or frustrated that you have been trying without success. Whether you are trying to get pregnant naturally or preparing for assisted reproductive treatments like IUI (intrauterine insemination) or IVF (in vitro fertilisation), nurturing the key ingredients will provide a foundation for the best chance of pregnancy success. What are they key ingredients? Quality eggs, healthy sperm, and a receptive uterus! Here are 5 tips to optimise your fertility for conception. Confirm you are OvulatingIf you are trying to conceive naturally, knowing when you ovulate helps you time intercourse to give yourself the best chance of success. You can monitor changes in body temperature or use at home testing kits to detect a surge in urine luteinising hormone but tracking these can be difficult, especially if you have PCOS. One of the best ovulation tracking techniques is cervical mucus monitoring. Under the influence of oestrogen, the water content of your cervical secretions increases and mucin content decreases. Cervical mucus monitoring can be a good predictor of ovulation. If your cervical mucus is very wet, stretchy and resembles raw egg white then ovulation is very close! This method of ovulation tracking is 74% accurate when compared to tracking ovulation via the gold standard of transvaginal ultrasound. There are four different types of cervical mucus.
Keep an eye out for type 4 cervical mucus. The odds of conception are at least 2 to 3 times higher when having sex on a day with type 4 cervical mucus than when having sex on a day with type 1 or 2 cervical mucus! Regular ovulation supports a healthy environment for your eggs to mature in. Even if you are undergoing IVF or IUI, keep track of this vital sign of ovulation as an indicator of egg health. Ovulating may also indicate that you are making enough progesterone to support a pregnancy. You can read more here about why ovulating is important and how to support healthy progesterone levels. Move Your BodyDid you know that exercise can improve the diversity of your gut microbiome for optimal fertility outcomes? Structured exercise can restore ovulation, reduce insulin resistance, improve sperm quality and DNA integrity and increase pregnancy and live birth rates. Exercise benefits include positive shifts in our gut microbiome to dampen inflammation, increase microbiota diversity and reduce the abundance of pathogenic bacteria. It also support optimal autonomic balance which is associated with IVF success. A snapshot of research study findings on fertility and exercise:
If you would like individualised guidance on the best exercise we are here to support you! It Takes Two!Male factors matter! Even if you are using a sperm donor to conceive sperm health still matters. It is estimated that in couples trying to conceive, male factors contribute to 50% of infertility. If you have PCOS, Endometriosis, Diminished Ovarian Reserve or Autoimmune condition then sperm health is even more important. Once sperm is stored and ready to fertilise an egg, it will not be able to repair any damage to DNA within the sperm that may be present. Sperm will always have some level of DNA damage but this is increased with increasing alcohol intake, insufficient or excessive physical activity, micronutrient deficiencies and poor gut health. So who takes on the role of repairing the sperm DNA? Our eggs! Yes, our eggs step up and get to work repairing fragmented DNA in the sperm. An egg can repair sperm with a few different pathways. It can use enzymes to fix methylation errors, and other processes to correct DNA sequences. Essentially the egg rolls up its sleeves and gets in there to pull things apart and put them back together so the sperm is functioning better. It takes a lot of energy to repair DNA damage in sperm. This requires our energy producing powerhouses in our eggs, our mitochondria, to work overtime. If the mitochondria content in our eggs is reduced, or the mitochondria just aren’t producing enough energy, our developing embryo runs out of steam. A typical egg may have 100,000 mitochondria. If you have PCOS, endometriosis, are not ovulating regularly or an imbalance in your gut microbiome you are likely to have less mitochondria, and these may not be producing as much energy. Nurturing your microbiome is a proven strategy to improve the health of mitochondria in your eggs, and to reduce DNA damage in sperm. Remember that it is a team effort! Micronutrients MatterThere are many important micronutrients to support preconception health and pregnancy but one you most likely have heard of is folate. Folate is one of the B group vitamins and is required for the development and growth of your baby. Folate is not stored as it is a water-soluble vitamin and any excess will be excreted in your urine, which is why you need to take a supplement. It is advised to start taking folate months before you decide you want to get pregnant. If your folate levels are low, a methylated folate which is more bioavailable may help your body absorb the folate more readily. Supplementing with methylated folate increases serum folate by 23% to 55% higher when compared to folic acid. Methylated folate may replete folate stores more uniformly and quickly. Ensure your prenatal meets your individual nutritional needs. Show Your Gut Some LoveOur gut microbiome influences the health of our eggs and sperm, and early pregnancy progression. An imbalance in our gut microbiome, or dysbiosis, may result from:
Dysbiosis, or disruption of the gut microbiota, is present in many conditions affecting fertility such as:
In a group of females experiencing recurrent implantation failure or multiple miscarriages, the diversity of their gut microbiome was reduced. Addressing this dysbiosis led to pregnancy rates that were 4 times higher. Even if you have experienced repeated failed rounds of IVF or ICSI, recurrent implantation failure or multiple pregnancy losses, we are here to help. References
Mena et al, 2019 Human Repro Update Duval et al 2015 Fertil Steril Hajizadeh et al, 2019 J Strength Cond Res ajizadeh et al, 2018 Life Sci Clark et al, 1995 Human Reprod Evans-Hoeker, et al., Fertil Steril, 2013 Bailey & Ayling, Sci Rep, 2018 A few days ago, my friend came to me with a question about corn. They had been told it was nutritionally redundant and were advised to drop it from their dietary repertoire. My friend wanted to know my thoughts on corn. I began by gently asking how they might feel about a life without barbecued corn cob, corn chips and guacamole, nachos, popcorn, and cornbread, not to mention the whimsical delight that is baby corn in stir fry. I think when we hear nutritional rhetoric that advocates the removal of any foods, let alone whole food groups, it is worth pausing to reflect how this will impact our well-being as a whole. In terms of nutrition, corn actually has a LOT going on. As do other yellow foods, which is the topic of this post. While I cannot hope to even scratch the surface of a whole plant colour in one post, I hope to inspire you to seek out these yello plant beauties to include them in ways that bring you enjoy. Since I’m already up on my high horse about corn, let’s start there… CornAs a starchy vegetable, corn is a rich source of carbohydrate, which is the most important fuel for our cells, particularly in the context of fertility. Corn is also a wonderful source of both soluble and insoluble fibres for healthy bowel movements, and prebiotic fibres to feed our gut bugs. Corn also contains lutein, a phytonutrient from the carotenoid family (which I introduced in the orange fruit and veg post). Lutein is famously known for its relationship to a lowered risk of age-dependent macular degeneration; however, it is also gaining attention as a potent antioxidant that can confer benefits to other organ systems, such as our cardiovascular system. Prebiotic BananaBanana is well-known for its rich potassium content which we need for nerve conduction, muscle contraction, and cell fluid balance. Bananas are also rich sources of important prebiotic fibres: inulin, pectin, and resistant starch (particularly green or just-ripe bananas), which are preferred foods of beneficial gut bugs such as Roseburia and Bifidobacteria species. In a study of 31 healthy women, 2 bananas a day for 60 days increased mean levels of Bifidobacteria species and were associated with lower levels of bloating (Mitsou et al. 2016). Bifidobacteria are a beneficial inhabitant of the gut. When they ferment prebiotic fibres they produce compounds that strengthen the intestinal barrier, reduce inflammation, balance hormones, and support your metabolism. All great news for fertility! PineapplePineapple contains a digestive enzyme called bromelain, which has been shown to increase the abundance of beneficial Akkermansia muciniphila in mice, as well as to support gut barrier integrity (Kostiuchenko, 2022). Akkermansia muciniphila is an important species in the gut, as it acts as a sentry in our mucus – eating just enough to prompt our colon cells to regenerate the mucus. Because bromelain is a digestive enzyme, it acts like a pair of scissors, chopping up nutrients into smaller nutrients so we can better absorb them. For this reason, bromelain has a cleansing effect on the gastrointestinal tract, contributing to good gastrointestinal health (Minich, 2019). Citrus Fruits for Fertility The inevitable eye-twitching, face-scrunching, cheek-sucking response to lemon juice is our body registering the citric acid and ascorbic acid (vitamin C). Combining acidic foods, such as citrus, with carbohydrate-rich foods, can help lower the rate at which our blood sugar spikes by 20-50%. Stable blood glucose levels are important for our cardiovascular health, as well as our fertility.I’m just in this moment reflecting on the wisdom of lemon juice + sugar on crepes… but I digress… It is not only the acid in citrus that make them a nutritional powerhouse for fertility. Introducing naringenin, an antioxidant found in citrus fruits. Naringenin has been observed to improve insulin sensitivity, metabolic disturbances, and inflammation (Mulvihill, Burke, & Huff, 2016). 8 weeks of naringenin treatment in PCOS rat models reduced ovarian damage, improved ovarian function, regulated sex hormone levels, and improved insulin sensitivity; it also increased the expression of occludin and claudin-1 (proteins crucial for intestinal barrier function and strength) and increased beneficial bacteria known to reduce inflammation via the production of short chain fatty acids (Wu et al, 2022). Naringenin has also been observed to ameliorate testicular toxicity in rats from Bisphenol A (BPA) (Alboghobeish, 2019). Effects of BPA on rats without naringin treatment included:
While the quantity administered in this study was in far higher doses than can be found from food (around the equivalent of ½ grapefruit per kg of body weight – so… 50 grapefruits a day for a 100kg male!), the exposure to BPA was far more concentrated too. Nobiletin is an anti-inflammatory flavonoid found in citrus fruit peel. Wei and colleagues (2018) administered nobiletin to mouse models of endometriosis and noted up to 67% reduction in endometriotic lesion size in mice given 20 mg/kg per day of nobiletin, as well as significantly reduced inflammation. Again, these doses are far higher than can be derived from consuming citrus fruit alone, and yet it is interesting that in an epidemiological study, an anti-endometriotic lesion effect was observed to track with increased citrus fruit intake (Harris, 2018). Finally, I want to tell you about rutin, yet another antioxidant found in citrus. When administered to rats, it was observed to prevent the development of endometriosis through antioxidant activity (Talebi et al, 2021). Rutin has also been observed to reduce oxidative stress and inflammation that contribute to testicular dysfunction (Rotimi, elebiyo, & Ojo, 2023). As a potent antioxidant, rutin has been observed to confer the following health benefits: anti-ageing (on skin); anti-neuro-degenerative; anti-hyperglycaemic; anti-dyslipidaemic; protective in liver damage and cardiovascular disorders. As if that’s not cool enough, when rutin is broken down (for example by our microbiota) the break-down products (Q-glc and Q) also confer potent health benefits. By consuming sources of rutin, we can increase the abundance of species that create these break-down products (Riva et al, 2020). Author: Emily Hahn, APD
References Alboghobeish S, Mahdavinia M, Zeidooni L, Samimi A, Oroojan AA, Alizadeh S, Dehghani MA, Ahangarpour A, Khorsandi L. Efficiency of naringin against reproductive toxicity and testicular damages induced by bisphenol A in rats. Iran J Basic Med Sci. 2019 Mar;22(3):315-523. doi: 10.22038/ijbms.2019.29757.7184. PMID: 31156794; PMCID: PMC6528718. Harris HR, Eke AC, Chavarro JE, Missmer SA. Fruit and vegetable consumption and risk of endometriosis. Hum Reprod. 2018 Apr 1;33(4):715-727. doi: 10.1093/humrep/dey014. PMID: 29401293; PMCID: PMC6018917. Ijaz, MU Tahir, A Samad, A Anwar, H (2021) Nobiletin ameliorates nonylphenol-induced testicular damage by improving biochemical, steroidogenic, hormonal, spermatogenic, apoptotic and histological profile. Human & Experimental Toxicology, 40(3), 403- 416 doi: 10.1177/0960327120950007 Kostiuchenko O, Kravchenko N, Markus J, Burleigh S, Fedkiv O, Cao L, Letasiova S, Skibo G, Fåk Hållenius F, Prykhodko O. Effects of Proteases from Pineapple and Papaya on Protein Digestive Capacity and Gut Microbiota in Healthy C57BL/6 Mice and Dose-Manner Response on Mucosal Permeability in Human Reconstructed Intestinal 3D Tissue Model. Metabolites. 2022 Oct 26;12(11):1027. doi: 10.3390/metabo12111027. PMID: 36355110; PMCID: PMC9696696. Minich, DM (2019) A Review of the Science of Colourful, Plant-Based Foods and Practical Strategies for Eating the Rainbow’, Journal of Nutrition and Metabolism, Volume 2019, Article ID 2125070. E.K. Mitsou, E. Kougia, Tz. Nomikos, M. Yannakoulia, K.C. Mountzouris, A. Kyriacou, Effect of banana consumption on faecal microbiota: A randomised, controlled trial, Anaerobe, Volume 17, Issue 6, 2011, Pages 384-387, ISSN 1075-9964, https://doi.org/10.1016/j.anaerobe.2011.03.018. Have you ever heard the phrase “Eat the Rainbow!”? In this article series we are going to explore what “eating the rainbow” means, the power of phytonutrients to create a fertile gut, and practical tips for how to eat the rainbow. We are going to welcome you to the vibrant world of orange fruits and vegetables! But first let’s find out more about the science of something that makes the rainbow possible: phytonutrients. If you want to see me talking all things orange you can join us in our Facebook group for the video! Introducing PhytonutrientsPhytonutrients are compounds found in plants that have effects on the body. Phyto: plant Nutrient: nourishment that is essential for the maintenance of life and growth There are thousands of phytonutrients, and these are pleiotropic, which means they can do more than one thing in our body. Phytonutrients can regulate inflammation, insulin sensitisation and ageing. They are powerful antioxidants that support cell signaling and messaging, epigenetic modulation of our DNA (i.e. altering which genes are expressed and when), and also interact with our gut microbiota. What’s the Rainbow got to do With Phytonutrients? Phytonutrients are the compounds in plants that confer colour. Using the phrase “eat the rainbow” points to the benefits of including a large variety of different coloured plants which will bring with them many different phytonutrients. Now, if we’re going to split hairs, the reality is that plants have multiple colour pigments in them. For example, pineapple contains the phytonutrients associated with green, purple, and orange fruits and veg. By eating a wide variety of fruits and vegetables, we are going to be getting many different benefits. ‘Eat the rainbow’ simply helps bring our awareness to just how many different types of fruit & veg variety we’re getting. Welcome to the World of Orange Fruits and Vegetables The major phytonutrient group found in orange fruits and vegetables are ‘carotenoids’. Our body use some of these to make vitamin A – an essential vitamin used by virtually every cell in the body, with important roles in embryonic development. Orange vegetables include root veg like carrots, sweet potatoes and yams, as well as veg that are technically fruits (since they have seeds): orange capsicum and pumpkin. Orange fruits include citrus fruits such as oranges, mandarins, tangerines; stone fruits such as peaches, nectarines, mangoes, apricots; and tropical fruits like persimmons, papayas, kumquats, passion fruits and rock melon. Orange Fruits & Vegetables and Your Fertility A study that followed over 1000 females for 12.5 years found that around 400mcg of beta-cryptoxanthin per day from orange-coloured fruits, including oranges, peaches, mandarins, may have the potential to delay ovarian senescence (ageing) by 1.3 years (Pearce & Tremellen, 2016). This data supports that orange fruits and vegetables provide essential antioxidant support for healthy reproductive function. In Nurses’ Health Study II (Harris et al, 2018) which had a study population of over 70,000 females, one serve of citrus fruits per day was associated with a lower risk of endometriosis risk compared to females who ate only 1 or less serving a week. Carotenoids like beta-carotene, found in citrus, increase the abundance of a species of bacteria called Faecalibacterium in rat models (Eroglu et al, 2022). Faecalibacterium is a key butyrate producing bacterial species. Butyrate has anti-inflammatory and gut-protecting properties, and has also been shown to reduce the size of endometriosis lesions in pre-clinical models (Chadchan et al., 2021). Carotenoids deliver fertility benefits for males too. Sperm is susceptible to DNA damage from increased reactive oxygen species and inflammation. Beta-carotene, which is found in carrots, pumpkin and oranges, has been associated with improved sperm parameters (Zareba et al, 2013). In another study, Beta-carotene was found in lower concentrations in the seminal fluid of infertile men when compared to fertile men (Palan & Naz, 1996). Orange fruits & Veg and our Gut Microbiome The positive benefits of carotenoids are likely, in part, due to the beneficial effects on our gut microbiome. Consuming plants rich in carotenoids has been shown to correlate with increased diversity of the gut microbiota, and this has been measured in large multi-ethnic study populations (Frankenfeld et al, 2021) as well as in pregnant women (Schmidt et al. 2021). Diversity of our gut microbiome supports ourthe production of fertility nurturing compounds. In addition to the phytonutrients in orange fruit and veg, we find prebiotic fibres including Fructooligosaccharides, Galactooligosaccharidesfructooligosaccharides, galactooligosaccharides, inulins, pectins, resistant starches… all delicious foods for our gut microbes. How to add Orange Fruits and Vegetables into Your Day To increase bioavailability, carotenoids from orange fruit and vegetables like to be in the presence of fat in our digestive tract. Here are some ideas for adding orange fruits and vegetables to your diet to support your reproductive health:
Adding orange fruits and vegetables to your diet is a simple way to improve your fertility and overall health. With a little planning, you can easily incorporate these nutrient-rich foods into your everyday meals. How are you going to add some orange fruits and vegetables to your week to support your reproductive health? Emily Hahn, APD References Chadchan, S.B., et al., Gut microbiota-derived short-chain fatty acids protect against the progression of endometriosis. Life Sci Alliance, 2021. 4(12). Eroglu A, Al'Abri IS, Kopec RE, Crook N, Bohn T. Carotenoids and Their Health Benefits as Derived via Their Interactions with Gut Microbiota. Adv Nutr. 2023 Mar;14(2):238-255. doi: 10.1016/j.advnut.2022.10.007. Epub 2022 Dec 16. PMID: 36775788. Frankenfeld, Cara & Hullar, Meredith & Maskarinec, Gertraud & Monroe, Kristine & Shepherd, John & Franke, Adrian & Randolph, Timothy & Wilkens, Lynne & Boushey, Carol & Marchand, Loïc & Lim, Unhee & Lampe, Johanna. (2021). The Gut Microbiome Is Associated with Circulating Dietary Biomarkers of Fruit and Vegetable Intake in a Multiethnic Cohort. Journal of the Academy of Nutrition and Dietetics. 122. 10.1016/j.jand.2021.05.023. H R Harris, A C Eke, J E Chavarro, S A Missmer, Fruit and vegetable consumption and risk of endometriosis, Human Reproduction, Volume 33, Issue 4, April 2018, Pages 715–727, https://doi.org/10.1093/humrep/dey014 Palan P, Naz R. Changes in various antioxidant levels in human seminal plasma related to immunoinfertility. Arch Androl. 1996 Mar-Apr;36(2):139-43. doi: 10.3109/01485019608987090. PMID: 8907675. Pearce K, Tremellen K. Influence of nutrition on the decline of ovarian reserve and subsequent onset of natural menopause. Hum Fertil (Camb). 2016 Sep;19(3):173-9. doi: 10.1080/14647273.2016.1205759. Epub 2016 Jul 18. PMID: 27430906. Schmidt KM, Haddad EN, Sugino KY, Vevang KR, Peterson LA, Koratkar R, Gross MD, Kerver JM, Comstock SS. Dietary and plasma carotenoids are positively associated with alpha diversity in the fecal microbiota of pregnant women. J Food Sci. 2021 Feb;86(2):602-613. doi: 10.1111/1750-3841.15586. Epub 2021 Jan 15. PMID: 33449409; PMCID: PMC10035785. Zareba P, Colaci DS, Afeiche M, Gaskins AJ, Jørgensen N, Mendiola J, Swan SH, Chavarro JE. Semen quality in relation to antioxidant intake in a healthy male population. Fertil Steril. 2013 Dec;100(6):1572-9. doi: 10.1016/j.fertnstert.2013.08.032. Epub 2013 Oct 2. PMID: 24094424; PMCID: PMC3843991. By Emily Hahn APD While dark green leafy vegetables may not always be a favourite at the dinner table, there is no denying the ‘eat your greens’ mealtime mantra is spoken as a gesture of love and good intention, founded in the wisdom that dark green and leafy vegetables are a rich source of body-loving nutrients. Let’s look at the humble vegetable group we call ‘dark green & leafy’ to see what all the fuss is about. Dark Green and Leafy VegetablesThis group of vegetables includes arugula (rocket), Bok choy (Chinese chard), collard greens (a variety of cabbage that produces long leaves instead of a head), dandelion greens, kale, mustard greens, rapini (broccoli raab), Swiss chard, turnip greens and spinach. It also includes the brassica vegetables since there are many nutritional overlaps. Brassicas (also called cruciferous vegetables) are broccoli, cauliflower, broccolini, turnip, Brussels sprouts, and cabbage. This short list is limited to those most cultivated in the West, but Mother Nature’s generosity in producing edible green plants the globe over means that many cultures have leafy greens native to their geographical location, and thus include them in their dietary pattern. Nutritionally, dark green and leafy vegetables are rich in:
Preparing for PregnancyWhile dark green and leafy vegetable consumption and fertility has not been heavily studied, research is consistent when it comes to the protective effects of dark green and leafy vegetables for positive birth outcomes and all-round health. Zerfu and colleagues (3) found that poor or inconsistent consumption of dark green leafy vegetables was associated with a 92% higher risk of experiencing an adverse pregnancy outcome compared to those with a higher or more consistent intake. Low or no green leafy vegetables consumption has also been identified as a risk factor for small for gestational age (4; 5), spontaneous preterm birth (4; 6), and gestational diabetes mellitus (4; 7). Low dark green and leafy vegetable intake has also been positively associated with anaemia during pregnancy, particularly in those with a low red meat consumption (8; 9). Finally, as rich sources of magnesium, dark green and leafy vegetables are important for preventing hypomagnesemia during pregnancy, which is associated with pre-eclampsia and pre-term birth (6). Adding Dark Green Leafy Vegetables to Your WeekSome nutrients in dark green leafy vegetables cannot be absorbed by the body in the absence of fat, so pairing these vegetables with a source of fat is a great way to ensure you’re getting all the nutritional benefits. Stuetz and colleagues (10) write about this beautifully in discussion of their findings that women in Tanzania who cooked their leafy vegetables with peanuts (a source of fat) were found to have higher levels of fat-soluble vitamin A in their blood. Here are some ideas for including dark green and leafy vegetables in your week:
Preparing for conception is a wonderful time to care for your body with the health-protective powers of dark green and leafy vegetables. If you’re looking for some further guidance on your fertility journey, we’d love to support you in clinic or via telehealth. You can book a complimentary phone call with us here or check out our science backed approach to get pregnant sooner in Create a Fertile Gut! References
In her absolute genius, mother nature has connected all non-essential bodily functions to physiological energy balance so in periods of famine non-essential physiological activity (such as reproduction) can be paused. While reproduction is of course necessary for survival of the species, the body prioritises individual survival… for the same reason flight attendants tell us to put on our own oxygen masks before we help others… that is… we’re no good to anyone, or our species, if we’re not functioning. Now, I appreciate that for many of us famine is a rare occurrence… I mean, how many of us live within a five-minute drive of at least 1 supermarket? Not to mention the abundance of corner shops and fast-food joints we pass on the way to aforementioned supermarket. Oh, and who has Uber Eats on their Smartphone? Yet, while we intellectually understand that we are not living in famine, our bodies and gut microbes base their definition of ‘famine’ on what passes through the gastrointestinal tract, and the regularity and consistency with which it arrives. So, on those weeks when we find ourselves too busy to schedule in regular times to eat, and it’s grab ‘n’ go at varying opportunistic times of the day (often making up for it in excess in the late evening) … the body is registering that we’re living in times of famine, and food is neither a predictable nor dependable commodity. The body feels safe shunting energy to fuel reproductive physiology when it knows there’s enough energy and sufficient nutritional diversity, delivered consistently. Let’s explore each of these… Energy AbundanceA reproductive cycle (from cultivating a viable egg, through pregnancy, to eventually weaning the offspring) is the most energetically costly activity female mammals undertake in their lifetime. Thus, the body has safe-keeping mechanisms in place to prevent risk to a mother or any potential offspring in times of caloric deficiency. In short – your beautiful body has totally got your back. Eating sufficient energy to match your metabolic activity is a must for fertility. Importantly, research suggests that it is not the absolute energy consumed that matters but that energy intake matches energy expenditure (1). Consuming insufficient energy for your needs may manifest as a lack of libido (suspected to be the result of disruption to ovarian steroid release, as well as reduced responsiveness to oestradiol by the part of the brain that controls female sexual behaviour), or manifest as suppressed ovulatory cycles (nil ovulation during your cycle or missing periods), due to a forebrain-controlled dampening of the luteinizing hormone (LH) surge that needs to occur for successful ovulation. This is why we often witness amenorrhea in females who are not meeting their energy expenditure requirements in the context of calorie restriction (e.g. dieting) or exercising without properly nutritionally replenishing themselves. An important caveat of energy abundance is that energy must be available for cells to use. For example, in the case of untreated diabetes, where there is an abundance of glucose trapped in the blood unable to be used as cellular energy, both sexual behaviours and ovulation are suppressed (1). If there is a risk that you may be living with untreated diabetes, getting help to manage blood glucose levels is important for your overall health, and will also increase your chances of falling pregnant. Similarly, in metabolic syndrome fatty acids are trapped in storage unable to be used for energy, hence infertility is a commonly observed side effect. Diversity AbundanceThe importance of diversity in the diet for fertility cannot be understated. Diversity (particularly of plants) not only ensures the availability of sufficient nutrients to help keep our human body functioning properly, it also ensures we are delivering a broad diversity of foods to our health-promoting gut microbes, who we know play an important role in supporting healthy sperm and egg development and uterine receptivity. A dietary pattern rich in different fruits, vegetables, fish, dairy, wholegrains, legumes, nuts, seeds, fish, poultry, and monounsaturated fats increases the probability of pregnancy following IVF (2, 3) and lowers the risk of ovulatory disorders and infertility due to other causes (4). If you would like to know if you are ovulating you can read here. Dietary patterns containing an abundance of different foods, rather than restrictive diets, positively correlate with biochemical pregnancy, clinical pregnancy, and probability of live birth (5). On the other hand, a Western Style Dietary pattern (low in plant diversity and healthy fats, while high in refined carbohydrates, vegetable oils, and processed meats) is associated with increased inflammation in follicular fluid and less blastocysts, as well as an increase in time to pregnancy, and delays in the female ovulatory cycle (6). Consistent AbundanceConsistent nourishment is a powerful signal to the body that the environment is conducive to reproduction. Indeed luteinizing hormone secretion (needed for ovulation) is tightly regulated by the forebrain (hypothalamus), which responds very rapidly to changes in availability of fuel (glucose or fatty acids) (1). Another note about consistency is the finding by Bavani and colleagues (7) that showed meal irregularity associated with irritable bowel syndrome prevalence. This is an interesting finding for fertility, since we know that IBS aligns with gut dysbiosis, which correlates with increased inflammation and oxidative stress, both of which negatively impact fertility. Consistently nourishing your body with a diverse array of foods sufficient in carbohydrate, protein, and health-promoting fats, rich in vitamins, minerals, phytochemicals, and antioxidants, is a wonderful first step when looking to conceive. If you are looking for support in cultivating nourishing practices for fertility, we would love to be there for you. Book in for a complementary call to see if we are the right support for you here. Blog Written by Emily Hahn APD References1. Wade, GN & Jones, JE (2004) Neuroendocrinology of nutritional infertility. Am J Physiol Regul Integr Comp Physiol 287: R1277–R1296, doi:10.1152/ajpregu.00475.2004.
2. Vujkovic M, de Vries JH, Lindemans J, et al. (2010) The preconception Mediterranean dietary pattern in couples undergoing in vitro fertilization/intracytoplasmic sperm injection treatment increases the chance of pregnancy. Fertil Steril. 94:2096–101. [PubMed: 20189169] 3. Twigt JM, Bolhuis ME, Steegers EA, et al. (2012) The preconception diet is associated with the chance of ongoing pregnancy in women undergoing IVF/ICSI treatment. Hum Reprod. 27:2526–31. [PubMed: 22593431] 4. Chavarro JE, Rich-Edwards JW, Rosner BA, Willett WC. (2007) Diet and lifestyle in the prevention of ovulatory disorder infertility. Obstetrics and gynecology. 110:1050–8. [PubMed: 17978119] 5. Sanderman, EA Willis, SK & Wise, LA (2022) Female dietary patterns and outcomes of in vitro fertilization (IVF): a systematic literature review, Nutrition Journal 21:5, https://doi.org/10.1186/s12937-021-00757-7 6. Skoracka, K Ratajczak, AE Rychter, AM Dobrowolska, A & Krela-Kazmlerczak (2021) Female Fertility and the Nutritional Approach: The Most Essential Aspects. Adv Nutr 2021;12:2372–2386; doi: https://doi.org/10.1093/advances/nmab068 7. Bavani, N.G., Hajhashemy, Z., Saneei, P. et al. (2022) The relationship between meal regularity with Irritable Bowel Syndrome (IBS) in adults. Eur J Clin Nutr 76, 1315–1322. https://doi.org/10.1038/s41430-022-01108-3 Your mental health impacts how your body functions. Short term stress can be beneficial for heightening your brain function, mounting immune responses to fight off colds and flu and to help you build resilience. It promotes positive adaptations. On the other hand, chronic stress can have negative consequences for your health and wellbeing, including your fertility. Psychosocial interventions to reduce stress and support individuals through fertility treatment have been found to double the chance of pregnancy (19). Let’s look at a practice that may help bring you calm, and support balance for a positive mind body connection. What is Mindfulness?Mindfulness is “paying attention to the present moment with openness, curiosity and without judgement” (1). Mindful awareness allows thoughts and experiences, often viewed through the lens of our opinions, preferences, and judgments, to simply be there. Some benefits of practising mindful awareness include:
his all sounds very nice, I hear you say, but what does this have to do fertility? Mindfulness Benefits Fertility and IVFDealing with infertility brings emotional and physical challenges. Mindfulness can help foster mental and emotional resilience, helping us to cope with feelings of anxiety, depression, distress, and worry (10). Research shows a correlation between mindfulness-based therapies and quality of life among women experiencing infertility (11-13), as well reduced rates of psychological distress, anxiety, and depression (13, 14). Li and colleagues (10) found that women who received mindfulness-based interventions alongside IVF treatments reported increased quality of life, self-compassion, and coping strategies for stress. They also found that these women had greater pregnancy rates compared to the group of women who did not receive the mindfulness-based intervention. How Do I Practice Mindfulness?There are two main types of mindfulness practice: formal mindfulness and informal mindfulness. Formal mindfulness Formal mindfulness meditation involves sitting or lying, with eyes closed, focusing the attention on one single thing. This could be the sensation of breathing, or focusing on sensations within the body (1). Daily formal mindfulness meditation has been correlated with decreased depression scores and enhanced sleep quality of women undergoing infertility treatments (15). Mindfulness apps, such as Smiling Mind, make it accessible to cultivate a regular mindful practice, offering free guided meditations that you can listen to at a time and place that suits you. You can also learn mindfulness from therapists trained in mindfulness-based therapies or attend classes where you can learn mindfulness practices as part of a group. Informal Mindfulness As with any skill, the more we practice mindfulness the more automatic it becomes. Informal mindfulness is about bringing the same level of focus and attention of formal mindfulness to aspects of your everyday life (1). That is, directing full, non-judgmental curiosity and attention to whatever it is you are doing in the present moment – be this commuting to work, vacuuming the house, eating, etc. Bringing mindful awareness to mealtimes can be a great way to cultivate regular mindful practice, providing opportunities to connect with your body and experience more deeply the sensory pleasure of appetite and eating. This increased attunement to the body puts us in a better position to nourish our body in a way that is aligned with what it needs in that moment. Because eating includes all 5 of our senses – seeing, smelling, tasting, touching, and hearing – it is a sensory rich context for focusing the attention. Next time you are eating a food, try bringing attention to how the food looks (colours, textures, shape), smells, the sound of picking it up with your utensil, and the mouth feel. Notice, without judgment, your response to the food, and how it feels in your body. Bring awareness to how this eating experience differs from the experience of, say, eating your lunch while checking your email, or snacking while in the car? Does your level of satisfaction improve? Yoga and FertilityYoga is a form of physical movement that marries up formal and informal mindfulness; the physical postures and breathing techniques of yoga are designed to bring mindful awareness to the body. A recent review found that anxiety scores were improved by yoga as an adjuvant during infertility treatment (16). Gaitzsch et al (17) also conducted a review of studies exploring the effectiveness of mind-body therapies (yoga and mindfulness-based therapies) during infertility treatment and found associations with lower anxiety and depression scores. Ready to Nurture Your Mind and Body?Cultivating a practice of mindfulness can help build resilience to life stress and enhance fulfilment in everyday experiences. Research suggests that incorporating mindfulness into your fertility journey can help reduce anxiety, depression, and distress, while also helping improve sleep and quality of life. If you would like to know more about incorporating mindful awareness in your everyday life, we would love to support you at The IVF Project. References1. Smiling Mind (2022) What is mindfulness? https://www.smilingmind.com.au/mindfulness.
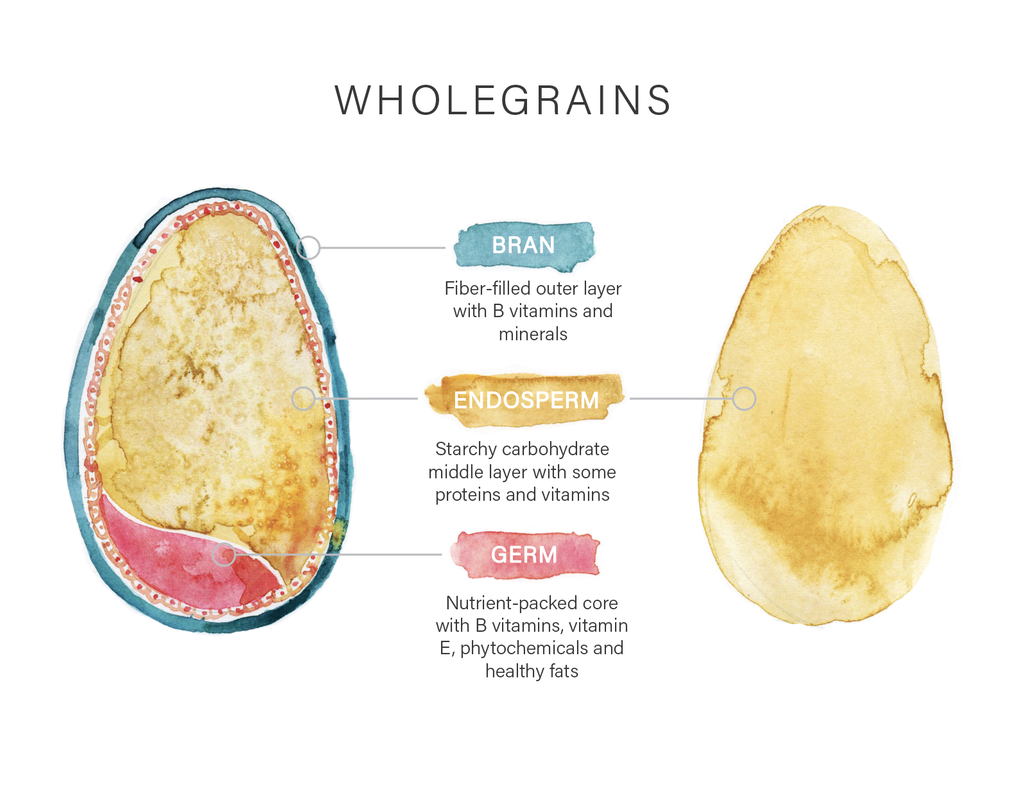
2. Chambers, R., Gullone, E., & Allen, N. B. (2009). Mindful emotion regulation: An integrative review. Clinical Psychology Review, 29(6), 560–572. https://doi.org/10.1016/j.cpr.2009.06.005 3. Lancaster, S., Klein, L., & Knightly, K. (2016). Mindfulness and Relaxation: a Comparison of Brief, Laboratory-Based Interventions. Mindfulness, 7(3), 614–621. https://doi.org/10.1007/s12671-016-0496-x 4. Luberto, C., Shinday, M., Song, N., Philpotts, R., Park, L., Fricchione, L., & Yeh, E. (2018). A Systematic Review and Meta-analysis of the Effects of Meditation on Empathy, Compassion, and Prosocial Behaviors. Mindfulness, 9(3), 708–724. https://doi.org/10.1007/s12671-017-0841-8 5. Rusch, H. L., Rosario, M., Levison, L. M., Olivera, A., Livingston, W. S., Wu, T., & Gill, J. M. (2019). The effect of mindfulness meditation on sleep quality: a systematic review and meta‐analysis of randomized controlled trials. Annals of the New York Academy of Sciences, 1445(1), 5–16. https://doi.org/10.1111/nyas.13996 6. Bartlett, L., Amanda, M A., Memish, N., Otahal, P & Kilpatrick M (2019). A Systematic Review and Meta-Analysis of Workplace Mindfulness Training Randomized Controlled Trials,.Journal of Occupational Health Psychology 24(1). 7. Dane, E., B. J. Brummel (2014). Examining workplace mindfulness and its relations to job performance and turnover intention. Human Relations, 67 (1), 105 - 128. 8. Blanck, P., Perleth, S., Heidenreich, T., Kröger, P., Ditzen, B., Bents, H., & Mander, J. (2018). Effects of mindfulness exercises as stand-alone intervention on symptoms of anxiety and depression: Systematic review and meta-analysis. Behaviour Research and Therapy, 102, 25–35. https://doi.org/10.1016/j.brat.2017.12.002. 9. Keng, S. L., Smoski, M. J., & Robins, C. J. (2011). Effects of mindfulness on psychological health: A review of empirical studies. Clinical psychology review, 31(6), 1041-1056. 10. Jing Li, Ling Long, Yu Liu, Wei He, Min Li, Effects of a mindfulness-based intervention on fertility quality of life and pregnancy rates among women subjected to first in vitro fertilization treatment, Behaviour Research and Therapy, Volume 77, 2016, Pages 96-104, ISSN 0005-7967, https://doi.org/10.1016/j.brat.2015.12.010. 11. Li, G., Jiang, Z., Han, X. et al. A moderated mediation model of perceived stress, negative emotions and mindfulness on fertility quality of life in women with recurrent pregnancy loss. Qual Life Res 29, 1775–1787 (2020). https://doi.org/10.1007/s11136-020-02460-2 12. Masoumeh Sadat Hosseini, Parvaneh Mousavi, Khadijeh Hekmat, Mohammad Hossein Haghighyzadeh, Reza Johari Fard, Razieh Mohammad Jafari. Effects of a short-term mindfulness-based stress reduction program on the quality of life of women with infertility: A randomized controlled clinical trial, Complementary Therapies in Medicine, Volume 50, 2020, 102403, ISSN 0965-2299, https://doi.org/10.1016/j.ctim.2020.102403. 13. Sherratt KA, Lunn S. Evaluation of a group programme of mindfulness-based cognitive therapy for women with fertility problems. J Obstet Gynaecol. 2013 Jul;33(5):499-501. doi: 10.3109/01443615.2013.786031. PMID: 23815205. 14. Xiaoran Wang & Yunxia Wang (2022) The Effectiveness of Mindfulness-Based Intervention on Emotional States of Women Undergoing Fertility Treatment: A Meta-Analysis, Journal of Sex & Marital Therapy, DOI: 10.1080/0092623X.2022.2109542 15. Cai-Feng Bai1,2, Nai-Xue Cui1, Xian Xu3, Guang-li Mi4, Ji-Wei Sun1, Di Shao5, Jie Li1, Yin-Zhi Jiang3, Qian-Qian Yang1, Xuan Zhang1, and Feng-Lin Cao1 Effectiveness of two guided self-administered interventions for psychological distress among women with infertility: a three-armed, randomized controlled trial. Human Reproduction, Vol.34, No.7, pp. 1235–1248, 2019 Advance Access Publication on June 26, 2019 doi:10.1093/humrep/dez066 16. Dumbala S, Bhargav H, Satyanarayana V, Arasappa R, Varambally S, Desai G, et al. Effect of yoga on psychological distress among women receiving treatment for infertility. Int J Yoga 2020;13:115-9. 17. Gaitzsch, H., Benard, J., Hugon-Rodin, J. et al. The effect of mind-body interventions on psychological and pregnancy outcomes in infertile women: a systematic review. Arch Womens Ment Health 23, 479–491 (2020). https://doi.org/10.1007/s00737-019-01009-8 18. Colleran, E (n.d.) What is mindfulness? https://blog.smilingmind.com.au/what-is-mindfulness. 19. Santa-Cruz, D.C., et al., Hair Cortisol Concentrations as a Biomarker to Predict a Clinical Pregnancy Outcome after an IVF Cycle: A Pilot Feasibility Study. Int J Environ Res Public Health, 2020. 17(9). There is one key diet component that is essential to fuel your fertility: carbohydrates! This may surprise you, as carbohydrates often get a bad rap. Reminiscent of the ‘low fat’ diets of the 80s and 90s, low carbohydrate diets such as Keto, Whole-30 and Paleo are touted as the panacea of health, promising to reduce inflammation and metabolic disruption. Reducing inflammation is positive for fertility, but is lowering carbohydrate the answer? What is Carbohydrate?Before we go any further, let’s just clear up what we mean by carbohydrate. Lots of the confusion surrounding carbohydrate stems from the fact that carbohydrates can be many different things. Carbohydrates are sugars, fibre and starches that can be found in a variety of foods from fruits and wholegrains to lollies. Carbohydrates are the preferred source of energy for your brain and muscles, and without them your gut microbes don’t have a food source to ferment to provide you with fertility loving metabolites like butyrate. The fine print: not all carbohydrates are created equal! We can categorise carbohydrates into two broad groups: simple and complex. Simple CarbohydratesSimple carbohydrates (also called simple sugars) are easily broken down by the body and the glucose is rapidly absorbed into the bloodstream. This produces a rapid and large blood sugar spike upon digestion. What goes up must come down, however, so following a blood sugar spike your body will also go through a relative low. A lot of these roller coaster peaks and troughs in blood sugar is correlated with inflammation in the body and the brain (1-3). Processed sources of simple carbohydrates often have the nutrients such as fibre, vitamins and minerals removed or reduced. You will recognise foods with this type of carbohydrate as they are high in energy, high in refined sugars (like white sugar, honey and fruit juice), high in refined grains (white flour), low in nutrients, low in fibre, high in sodium content and could be high in saturated fat, cholesterol and trans fats. Essentially you get less nutritional bang for your buck with simple carbohydrates. Examples of simple carbohydrates are sugar, honey, agave nectar, maple syrup, high fructose corn syrup, rice malt syrup, refined (white) flour, fruit juice, soft drinks, biscuits, lollies, cakes, chips, sugary breakfast cereals, white bread and wraps. Reducing your intake of refined sources of simple carbohydrate is a win for your fertility. Complex CarbohydratesComplex carbohydrates are made up of more complex sugars strung together in long chains. Compared to simple carbohydrates, foods with complex carbohydrates are packed with more nutrients, vitamins, minerals and fibre. This beneficial carbohydrate is often found in whole foods - foods that have not been overly processed or turned into something that you don’t recognise anymore. Whole foods with complex carbohydrates (fibre and starches) include: wholegrains, brown rice, vegetables, fruit, nuts and seeds, beans and legumes. You will recognise them as they contain low to moderate energy, have a high nutrient content, have no refined sugars or grains, are naturally high in fibre, low in sodium (salt) and have low to no saturated and trans fats. Fibre and starch are two types of complex carbohydrates. Starches are naturally occurring in breads, cereals, starchy vegetables (peas, corn, beans, and potatoes), legumes and wholegrains. Starches will be found with fibre, although some foods have more fibre than starch. Fibre is naturally found in fruits, vegetables, legumes, nuts, seeds and wholegrains. Complex carbohydrates feed beneficial bacteria in your gut microbiota that can enhance your fertility by lowering inflammation, increasing your gut diversity, and reducing dysbiosis. To achieve a fertile gut you need to include complex carbohydrates in your diet that will feed those bacteria who are responsible for looking after you. They can’t do it without you! Wholegrains – a Whole Lotta GoodnessWholegrains are one of the most important carbohydrates for fertility. In Australia, wholegrains are grain foods that have not been processed or refined. Wholegrains have not had their outer bran or germ layers removed, and therefore contain an abundance of nutrients that support fertility. They contain more fibre, vitamins, minerals and antioxidants than their refined versions (e.g. white bread). Wholegrain intake has been associated with better antioxidant defenses and reductions in inflammation, as well as better blood glucose regulation (good news for polycystic ovarian syndrome (PCOS)-related insulin resistance) (4). Wholegrain consumption has also been shown to increase the production of fertility-promoting compounds by the gut microbiota (5) and a higher preconception intake of wholegrains has been associated with thicker endometrium for implantation and increased live birth rates following IVF (4). Wholegrain examples include wholewheat, rice (brown, black), corn (including popcorn), oats, barley, spelt, rye, sorghum, millet and pseudo grains such as buckwheat, quinoa, wild rice and amaranth. Wholegrains Reduce InflammationWholegrain foods are not only great for fertility. They have a positive impact on reduced risk of developing type 2 diabetes, certain type of cancers and cardiovascular diseases. Wholegrain intake is associated with a reduction in circulating inflammatory markers like CRP (6). Higher CRP levels can be detrimental to the outcome of assisted reproductive treatments (7) while reducing inflammation improves egg and sperm health and uterine receptivity. Are you Eating Enough Wholegrains?How many servings of wholegrains are you getting per day? One serve of wholegrains might look like:
|
|
1300 084 694
[email protected] Suite T36, 477 Boundary St Spring Hill, Brisbane QLD 4000 Fax. 07 3540 8164 Copyright © 2022
|